 SHEFA Medical
SHEFA Medical
 SHEFA Medical
SHEFA Medical
In today’s healthcare landscape, understanding "How To Evaluate Medical Product Quality For Bulk Purchasing" is crucial. Dr. Emily Carter, a renowned expert in medical supply evaluation, emphasizes, "Quality assessment is not just a step; it’s a commitment to patient safety." As institutions consider bulk buying, the quality of medical products becomes paramount.
Effective evaluation starts with reliable sources. Look for certifications from regulatory bodies. These certifications indicate adherence to safety and quality standards. However, documents can be misleading. Always verify claims through direct inquiries with manufacturers. Collecting feedback from other healthcare providers is another effective way. Their experiences can guide your decisions.
Testing samples before bulk purchasing can provide clear insights. It mitigates risks associated with poor quality. Regular audits of the suppliers also maintain ongoing quality assurance. The complexity of evaluating medical products cannot be overstated. Each step requires scrutiny, and adjustments may be necessary along the way.
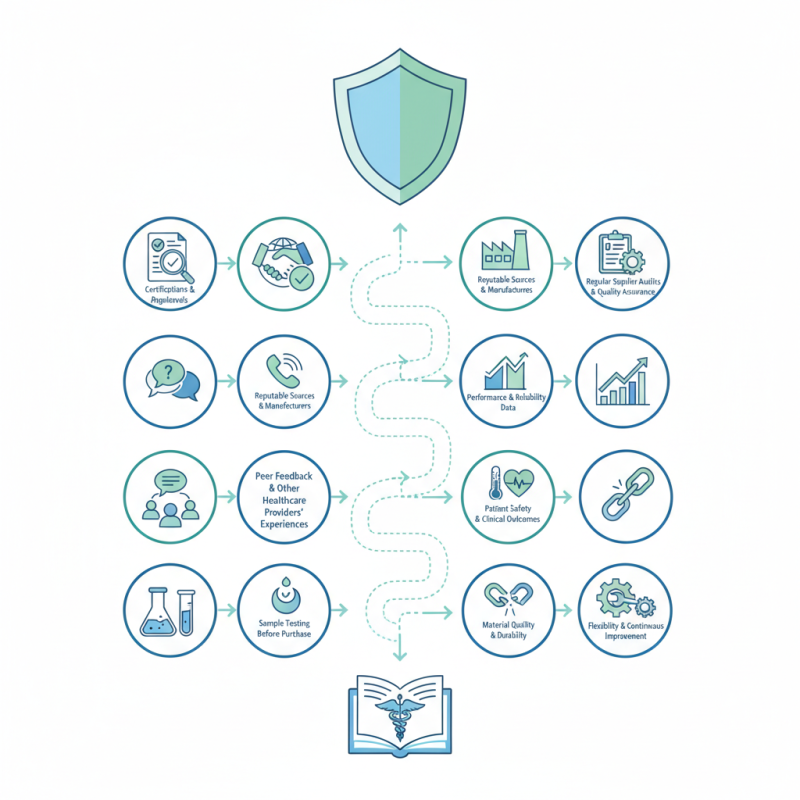
When evaluating the quality of medical products for bulk purchasing, identifying key quality standards is crucial. Certifications such as ISO 13485 indicate compliance with international quality management systems. Products bearing these certifications often meet safety and efficacy benchmarks. However, verifying the authenticity of these certifications can sometimes be challenging.
Pay attention to material specifications. These specifications govern the safety and performance of medical products. For instance, biocompatibility is vital in products that contact human tissue. Ensuring a product is made from approved materials can significantly reduce risks. In addition, traceability of components is often overlooked. Understanding where materials come from adds another layer of accountability.
Consider also the manufacturer’s history. A company's track record for quality can reveal much about their reliability. Look for testimonials or case studies. However, not all information may present a complete picture. Reflecting on past experiences can help gauge future performance. Each product evaluation should include a thorough review of documentation and supplier interactions. These steps can help you make informed decisions.
When purchasing medical products in bulk, understanding regulatory compliance is essential. Regulatory bodies set strict standards to ensure product safety and efficacy. Compliance guarantees that the products have been tested and meet necessary quality criteria. This can significantly reduce risks in bulk buying.
It's crucial to evaluate if the manufacturer adheres to Good Manufacturing Practices (GMP). Look for certifications that represent compliance with industry standards. Ask for documentation that proves their quality control processes. Many companies may present attractive prices, but savings should not compromise quality. A reliable supplier will provide transparent information on their regulatory adherence.
One tip is to conduct audits or inspections of the manufacturing facility. This hands-on approach allows for a better understanding of their processes and commitment to regulation. Additionally, maintain clear communication with suppliers about their compliance status. Sharing expectations can help establish a trustworthy relationship. Remember that understanding regulatory compliance can prevent costly mistakes down the line. Each decision requires careful consideration to ensure patient safety is prioritized.
When evaluating medical product quality for bulk purchasing, supplier certifications are crucial. According to the World Health Organization (WHO), approximately 10% of medical products are substandard or falsified. This is a significant concern, as such products can threaten patient safety and healthcare integrity. Therefore, assessing certifications like ISO 13485 can provide insight into a supplier’s commitment to quality management in the medical device field.
Quality assurance processes also play a vital role. A report by the Institute of Medicine indicates that rigorous quality checks can reduce errors by up to 50%. It is essential to inquire about the supplier’s quality control protocols. Do they conduct internal audits? What are their procedures for handling defective products? These factors indicate their overall operational integrity.
Nonetheless, some suppliers may lack transparency. A 2022 survey highlighted that 30% of healthcare providers reported difficulties in obtaining certification information from suppliers. This gap raises questions about reliability. Buyers must remain vigilant in verifying all claims made by suppliers. Engaging with industry professionals can offer insights into effective evaluation strategies. A continuous effort towards supplier assessment is necessary to ensure that high-quality medical products are acquired safely.
When bulk buying medical products, comprehensive testing and validation are crucial. Quality varies significantly across suppliers. Industry reports indicate that nearly 30% of medical products in bulk do not meet safety standards. Each product must undergo rigorous testing to ensure it meets established guidelines.
Validation processes should include stability testing, performance assessments, and adverse event monitoring. A 2022 survey revealed that only 55% of healthcare providers conduct thorough assessments before procurement. This gap can pose significant risks to patients and healthcare institutions. Validate sourcing quality through certifications or third-party testing results whenever possible.
Documentation is key. Keeping detailed records of product testing and validation helps ensure compliance and traceability. Poor documentation can lead to lapses in accountability, raising questions about product safety. Regular audits and reviews of quality practices should be the norm, yet only 40% of organizations commit to this rigorously. Addressing these issues can enhance confidence in bulk medical products.
| Evaluation Criteria | Description | Importance Level | Testing Methods |
|---|---|---|---|
| Regulatory Compliance | Ensure products meet local and international regulatory standards. | High | Document Review, Certification Checks |
| Quality Assurance | Assessment of the manufacturer’s quality management system. | High | Audits, ISO Certification Review |
| Material Safety | Analysis of materials used in production for safety and efficacy. | Medium | Lab Testing, Toxicology Reports |
| Performance Testing | Evaluation of product performance against specifications. | High | Functionality Tests, User Trials |
| Shelf Life Testing | Determining the product's shelf stability and usability over time. | Medium | Accelerated Aging Tests, Storage Studies |
| Packaging Evaluation | Examination of packaging integrity and protection during transport. | Medium | Packaging Stress Tests, Environmental Impact Assessments |
| User Feedback | Incorporating input from healthcare professionals and end-users. | High | Surveys, Focus Groups |
| Cost-Benefit Analysis | Evaluating the cost in relation to the quality and performance. | High | Financial Review, Comparative Analysis |
| After-Sales Support | Evaluating the support available post-purchase for maintenance. | Medium | Service Agreements, Customer Service Evaluations |
| Documentation and Traceability | Ensuring complete and accurate documentation for traceability. | High | Document Audits, Compliance Checks |
When it comes to bulk buying medical products, evaluating user feedback and product performance data is crucial. User feedback provides insights that go beyond superficial marketing claims. It reflects real experiences and can reveal potential issues. Look for patterns in reviews. If multiple users report a similar problem, take note. This can help you make informed decisions.
Analyze product performance data as well. Metrics like defect rates and return statistics are invaluable. High-quality products generally have low return rates and positive reviews. Compare these figures across similar products. This comparison helps you understand how one product stands against its competitors.
Gather detailed user experiences. This can highlight strengths and weaknesses. Be cautious of overly positive reviews. Sometimes, they are too good to be true. Seek feedback from various sources, like forums and professional reviews. Remember that every product has its imperfections. Acknowledging potential shortcomings is part of a comprehensive evaluation process. What works well for one user may not suit another's needs. Keeping an open mind will lead to better choices.
: Regulatory compliance ensures product safety and efficacy. It reduces risks when purchasing in bulk.
GMP are guidelines that manufacturers must follow to ensure quality and safety in production.
Request documentation and certifications. Transparency is key in confirming their adherence to regulations.
Certifications indicate a supplier's commitment to quality. They help assure that products meet necessary standards.
Audits provide insights into a supplier's processes and regulatory commitment. It enhances trust in their operations.
Some suppliers may not share certification information. This can complicate the verification process for buyers.
Roughly 10% of medical products may be substandard. This poses risks to patient safety and healthcare integrity.
Compromising quality for cheaper options can lead to costly mistakes and jeopardize patient safety.
Inquire about their internal audits and how they handle defective products.
Engage with industry professionals. Continuous effort in evaluation helps ensure high-quality products.
In the process of bulk purchasing medical products, understanding how to evaluate medical product quality is crucial. Key quality standards must first be identified, ensuring that products meet both industry benchmarks and effectiveness requirements. Compliance with regulatory manufacturing guidelines is vital to guarantee safety and reliability, and thorough scrutiny of suppliers’ certifications will provide insights into their quality assurance processes.
Additionally, conducting comprehensive testing and validation of products is essential to assess their performance in real-world scenarios. Evaluating user feedback and performance data can further inform purchasing decisions, ensuring that the selected medical products not only meet set standards but also fulfill the needs of end-users. By focusing on these strategies, buyers can make informed decisions that prioritize quality in their bulk purchasing initiatives.
